One hundred years ago this month, in Feb. 1924, a hereditary member of the French nobility, Louis Victor Pierre Raymond, the 7th Duc de Broglie, published a landmark paper in the Philosophical Magazine of London [1] that revolutionized the nascent quantum theory of the day.
Prior to de Broglie’s theory of quantum matter waves, quantum physics had been mired in ad hoc phenomenological prescriptions like Bohr’s theory of the hydrogen atom and Sommerfeld’s theory of adiabatic invariants. After de Broglie, Erwin Schrödinger would turn the concept of matter waves into the theory of wave mechanics that we still practice today.

The story of how de Broglie came to his seminal idea had an odd twist, based on an initial misconception that helped him get the right answer ahead of everyone else, for which he was rewarded with the Nobel Prize in Physics.
de Broglie’s Early Days
When Louis de Broglie was a student, his older brother Maurice (the 6th Duc de Broglie) was already a practicing physicist making important discoveries in x-ray physics. Although Louis initially studied history in preparation for a career in law, and he graduated from the Sorbonne with a degree in history, his brother’s profession drew him like a magnet. He also read Poincaré at this critical juncture in his career, and he was hooked. He enrolled in the Faculty of Sciences for his advanced degree, but World War I side-tracked him into the signal corps, where he was assigned to the wireless station on top of the Eiffel Tower. He may have participated in the famous interception of a coded German transmission in 1918 that helped turn the tide of the war.
Beginning in 1919, Louis began assisting his brother in the well-equiped private laboratory that Maurice had outfitted in the de Broglie ancestral home. At that time Maurice was performing x-ray spectroscopy of the inner quantum states of atoms, and he was struck by the duality of x-ray properties that made them behave like particles under some conditions and like waves in others.


Through his close work with his brother, Louis also came to subscribe to the wave-particle duality of x-rays and chose the topic for his PhD thesis—and hence the twist that launched de Broglie backwards towards his epic theory.
de Broglie’s Massive Photons
Today, we say that photons have energy and momentum although they are massless. The momentum is a simple consequence of Einstein’s special relativity

And if m = 0, then
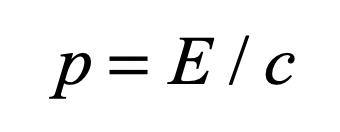
and momentum requires energy but not necessarily mass.
But de Broglie started out backwards. He was so convinced of the particle-like nature of the x-ray photons, that he first considered what would happen if the photons actually did have mass. He constructed a massive photon and compared its proper frequency with a Lorentz-boosted frequency observed in a laboratory. The frequency he set for the photon was like an internal clock, set by its rest-mass energy and by Bohr’s quantization condition

He then boosted it into the lab frame by time dilation
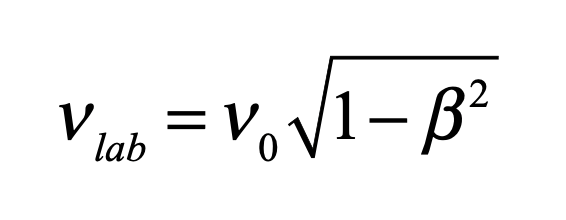
But the energy would be transformed according to

with a corresponding frequency

which is in direct contradiction with Bohr’s quantization condition. What is the resolution of this seeming paradox?
de Broglie’s Matter Wave
de Broglie realized that his “massive photon” must satisfy a condition relating the observed lab frequency to the transformed frequency, such that

This only made sense if his “massive photon” could be represented as a wave with a frequency
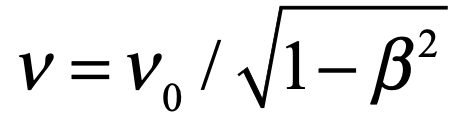
that propagated with a phase velocity given by c/β. (Note that β < 1 so that the phase velocity is greater than the speed of light, which is allowed as long as it does not transmit any energy.)
To a modern reader, this all sounds alien, but only because this work in early 1924 represented his first pass at his theory. As he worked on this thesis through 1924, finally defending it in November of that year, he refined his arguments, recognizing that when he combined his frequency with his phase velocity,
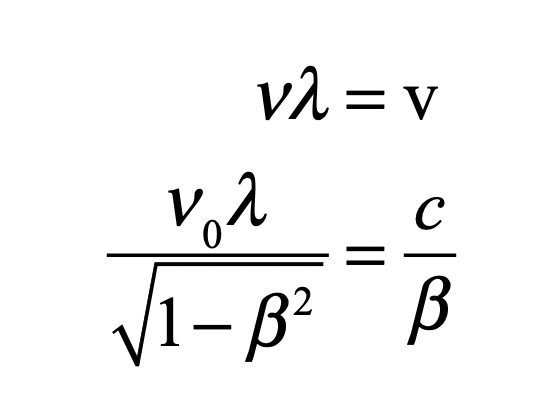
it yielded the wavelength for a matter wave to be

where p was the relativistic mechanical momentum of a massive particle.
Using this wavelength, he explained Bohr’s quantization condition as a simple standing wave of the matter wave. In the light of this derivation, de Broglie wrote
We are then inclined to admit that any moving body may be accompanied by a wave and that it is impossible to disjoin motion of body and propagation of wave.
pg. 450, Philosophical Magazine of London (1924)
Here was the strongest statement yet of the wave-particle duality of quantum particles. de Broglie went even further and connected the ideas of waves and rays through the Hamilton-Jacobi formalism, an approach that Dirac would extend several years later, establishing the formal connection between Hamiltonian physics and wave mechanics. Furthermore, de Broglie conceived of a “pilot wave” interpretation that removed some of Einstein’s discomfort with the random character of quantum measurement that ultimately led Einstein to battle Bohr in their famous debates, culminating in the iconic EPR paper that has become a cornerstone for modern quantum information science. After the wave-like nature of particles was confirmed in the Davisson-Germer experiments, de Broglie received the Nobel Prize in Physics in 1929.
Louis de Broglie was clearly ahead of his times. His success was partly due to his isolation from the dogma of the day. He was able to think without the constraints of preconceived ideas. But as soon as he became a regular participant in the theoretical discussions of his day, and bowed under the pressure from Copenhagen, his creativity essentially ceased. The subsequent development of quantum mechanics would be dominated by Heisenberg, Born, Pauli, Bohr and Schrödinger, beginning at the 1927 Solvay Congress held in Brussels.

By David D. Nolte, Feb. 14, 2024
[1] L. de Broglie, “A tentative theory of light quanta,” Philosophical Magazine 47, 446-458 (1924).



[…] 100 Years of Quantum Physics: de Broglie’s Wave (1924) […]
LikeLike